The Hong Research Group works at the interface of materials science, biology, and nanotechnology to develop novel polymeric nanodevices for biological analysis, diagnostics, and therapeutics.
Core Focus Areas
This is an accordion element with a series of buttons that open and close related content panels.
Novel Nanocarrier Platforms
Dendritic nanoparticles for enhanced binding kinetics in immunotherapy
- Immune checkpoint inhibitors are increasingly used in cancer treatment regimens, but still have a low success rate. One path to increased efficacy is increasing the binding strength of the inhibitor molecule. Dendrimer nanoparticles provide a route to increased binding strength through increased avidity by multivalent interactions. We have developed poly(amidoamine) dendrimer nanoparticles functionalized with antibodies and peptides to enhance PD-L1 blockade therapies, providing a novel platform for cancer immunotherapy.
- Related Publications:
Dendrimer-exosome hybrid nanoparticles for drug delivery
- Many nanomedicine formulations fail to reach the clinic due to immunogenicity of the delivery system or undesirable biodistributions. We have developed techniques to combine the advantages of dendrimer nanoparticles as drug delivery platforms with exosomes to enhance immune interactions and systemic delivery. The exosome-dendrimer hybrid nanoparticles have shown enhanced delivery of oligonucleotide therapies with lower cytotoxicity.
Dendron micelles for combination therapy
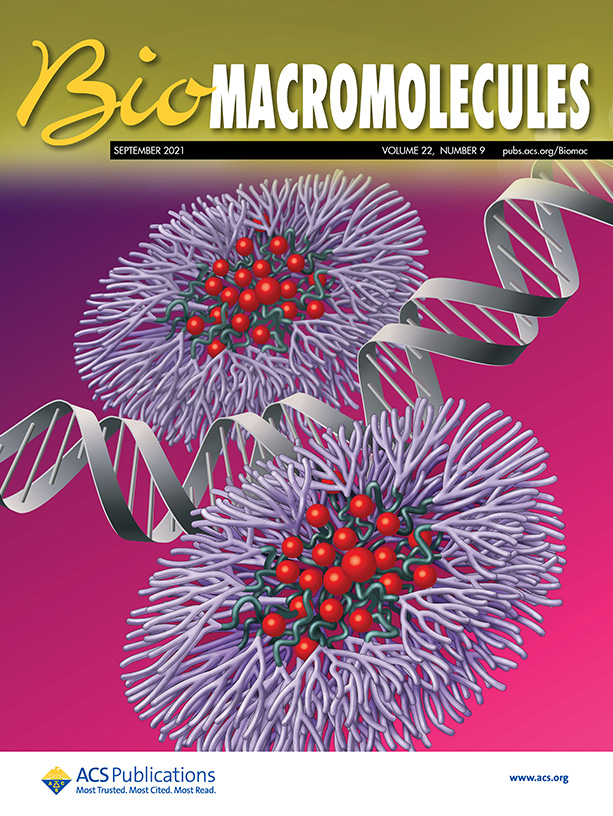
- The use of two or more therapeutic agents is increasing as the standard of care in treating many cancers. Multiple therapeutics can be delivered together, with controlled release, by engineered nanomedicines. Using our basic understanding of nano-bio interactions we have prepared polymeric nanoparticles combining advantageous properties of multiple systems and currently applying them towards combination therapy for cancer treatment applications. Micelles self-assembled from linear and dendritic amphiphilic copolymers represent a promising class of nanocarriers. We have developed dendron-based micelles consisting of a hydrophobic, biodegradable poly(ε-caprolactone) core, a hyperbranched generation 3 polyester dendrimer, and a poly(ethylene glycol) outer layer. Owing to the hyperbranched dendron structure, our micelles demonstrated enhanced thermodynamic stability, a high degree of surface PEG coverage, and are able to maintain ligand-mediated cell targeting in the presence of serum proteins. The dendron micelles further allow for control over the surface distribution of targeting ligands.
- Related Publications:
Liquid Biopsy
Highly sensitive circulating tumor cell isolation for liquid biopsy
- Metastatic cancer is initiated with an adhesion mechanism hijacked from the inflammation response. The first step in this process involves transient, reversible, adhesive interactions between selectin molecules expressed on the endothelial venules and glycoprotein receptors on the leukocyte/invasive cancer cell, resulting in cell rolling. In the second step, circulating tumor cells (CTCs) are held in place by cancer cell-specific ligands. By attaching those ligands to dendrimer nanoparticles, we encourage multivalent binding and achieve exceptional sensitivity. We have shown that the surface can be tuned to target specific cancers and currently have multiple clinical studies exploring CTC enumeration as a tool to predict clinical outcomes following treatment.
- Related Publications:
Tumor-derived exosome isolation for liquid biopsy
- Exosomes are released by tumors in larger quantities than circulating tumor cells and may represent a valuable biomarker for cancer treatment and progression. However, the small size and buoyancy of exosomes makes their isolation a challenge. We have developed surfaces with multiple layers of dendrimers capable of achieving highly sensitive, tumor-specific exosome capture and confirmed that the process is due to enhanced multivalent binding at the nanoscale by cancer-specific ligands on dendrimer nanoparticles. Additional work is exploring the use of multi-modal liquid biopsies for improved clinical accuracy in liquid biopsy.
- Related Publications:
Nano-bio Interactions
Nanoparticle-cell interactions and nanoparticle-tissue interactions
- Related research project: Developing a Fundamental Understanding of Nano-Bio Interactions
Current Projects
Milestones and Achievements
![]() Aug. 2020: Our postdoc, Dr. Woo-jin Jeong, accepted an offer from Inha University in Korea to join its Biomedical Engineering faculty as an Assistant Professor as of Sept. 1. This great achievement makes him the third university faculty from our lab! Congratulations Woo-jin!!!
Aug. 2020: Our postdoc, Dr. Woo-jin Jeong, accepted an offer from Inha University in Korea to join its Biomedical Engineering faculty as an Assistant Professor as of Sept. 1. This great achievement makes him the third university faculty from our lab! Congratulations Woo-jin!!!
![]() June 2020: Our paper entitled “An Avidity-based PD-L1 Antagonist Using Nanoparticle-Antibody Conjugates for Enhanced Immunotherapy” has been accepted for publication by Nano Letters. The authors are Jiyoon, Ashita, Mari, Woo-jin, Mike, Kara, Luke, Elizabeth, Dr. Wheeler, and Dr. Hong. Congratulations!
June 2020: Our paper entitled “An Avidity-based PD-L1 Antagonist Using Nanoparticle-Antibody Conjugates for Enhanced Immunotherapy” has been accepted for publication by Nano Letters. The authors are Jiyoon, Ashita, Mari, Woo-jin, Mike, Kara, Luke, Elizabeth, Dr. Wheeler, and Dr. Hong. Congratulations!
![]() June 2020:Our paper entitled “An Avidity-based PD-L1 Antagonist Using Nanoparticle-Antibody Conjugates for Enhanced Immunotherapy” has been accepted for publication by Nano Letters. The authors are Jiyoon, Ashita, Mari, Woo-jin, Mike, Kara, Luke, Elizabeth, Dr. Wheeler, and Dr. Hong. Congratulations!
June 2020:Our paper entitled “An Avidity-based PD-L1 Antagonist Using Nanoparticle-Antibody Conjugates for Enhanced Immunotherapy” has been accepted for publication by Nano Letters. The authors are Jiyoon, Ashita, Mari, Woo-jin, Mike, Kara, Luke, Elizabeth, Dr. Wheeler, and Dr. Hong. Congratulations!
Journal Covers