
Assistant Professor Quanyin Hu and his CIPT Lab team addresses one of the biggest limitations of treating disease with probiotics
By Eric Hamilton
This article originally appeared on UW–Madison’s news page.
Scientists studying probiotics, beneficial bacteria that show promise for their ability to treat inflammatory bowel disease and other intestinal disorders, continue to face a problem: how to keep probiotics from getting obliterated in the gut before they can be helpful.
Researchers at the University of Wisconsin–Madison School of Pharmacy think they have a solution involving a dual-layer protection system that can keep probiotic bacteria alive in the lower intestine long enough to help treat or prevent colitis in a mouse model of the disease.
Quanyin Hu and postdoctoral researcher Jun Liu have filed for a patent on their double-protection technique and, with their collaborators, published their findings Sept. 26 in the journal Nano Today.
If the new technique shows similar effectiveness in clinical trials, it could help advance the use of probiotics.
The research addresses one of the biggest limitations of treating disease with probiotics, which are bacteria that promote healthy tissue development and improve the gut microbiome.
“When you transfer these bacteria through the oral route, most of them are getting killed by the acidic environment of the stomach. Or they’re getting cleared out of the intestine because they aren’t adhering,” says Hu, the senior author of the report. “Our double protection technique addresses these limitations.”
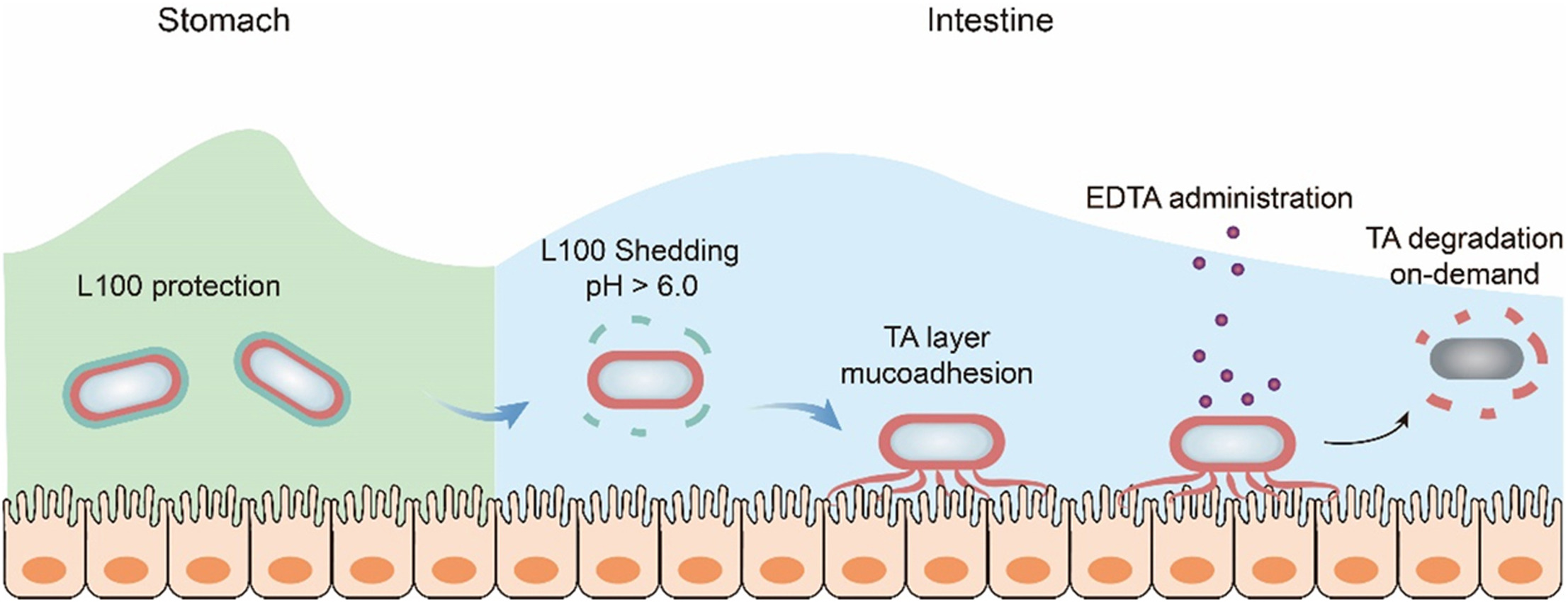
To protect against stomach acid, the researchers coated a probiotic strain of E. coli bacteria first in tannic acid, then in a polymer called L100. Tannic acid is a common food additive. Since it’s sticky, the researchers selected it to help the bacteria hold onto the lining of the intestines so they aren’t cleared by the body as quickly. L100, already used in extended-release drug formulations, is stable in acidic environments but dissolves in the less-acidic environment of the intestine.
Together, this produced a protective shell that, when fed to mice, increased the number of bacteria found in their intestines and how long they stuck around.
The team tested their new strategy in a mouse model of colitis, an inflammation of the lower intestine. Researchers can induce the condition by feeding the animals a disease-causing chemical. When used to treat already-established colitis, the mice fed double-coated bacteria lost less weight and had healthier intestines than mice given uncoated bacteria or none at all.
Going forward, the researchers plan to test the method’s safety and effectiveness in other animal models on the way to potential human clinical trials.
The researchers also tested the ability of the protected probiotics to prevent the formation of colitis by providing them to mice before adding in the disease-inducing chemical. Again, the double-coated probiotics helped mice maintain their weight better than when they were provided uncoated bacteria, although the treatment did not entirely prevent weight loss and other signs of disease.
Hu’s team considered one potential side effect of their new technique — the tannic acid is so sticky, it could coat the intestine for too long. “This adherence could cause side effects by preventing the absorption of nutrients,” says Hu.
So, the researchers tested a method of washing away the tannic acid and bacteria after the treatment was done. When they fed mice the nontoxic chemical EDTA, which interferes with tannic acid’s stickiness, the scientists saw that the probiotic bacteria were cleared by the intestines faster, as they hoped.
Going forward, the researchers plan to test the method’s safety and effectiveness in other animal models on the way to potential human clinical trials.
“I think if our project can be transferred into the clinic it will be very valuable and very useful,” says Liu.
Learn more about the Hu Cell Inspired Personalized Therapeutic (CIPT) Lab.

Gel Loaded with Cancer-Fighting Cells Keeps Tumors in Check After Surgery in Mice
Read about Dr. Hu's work on an innovative technique to deploy the immune system to wipe up cancer cells after surgery.