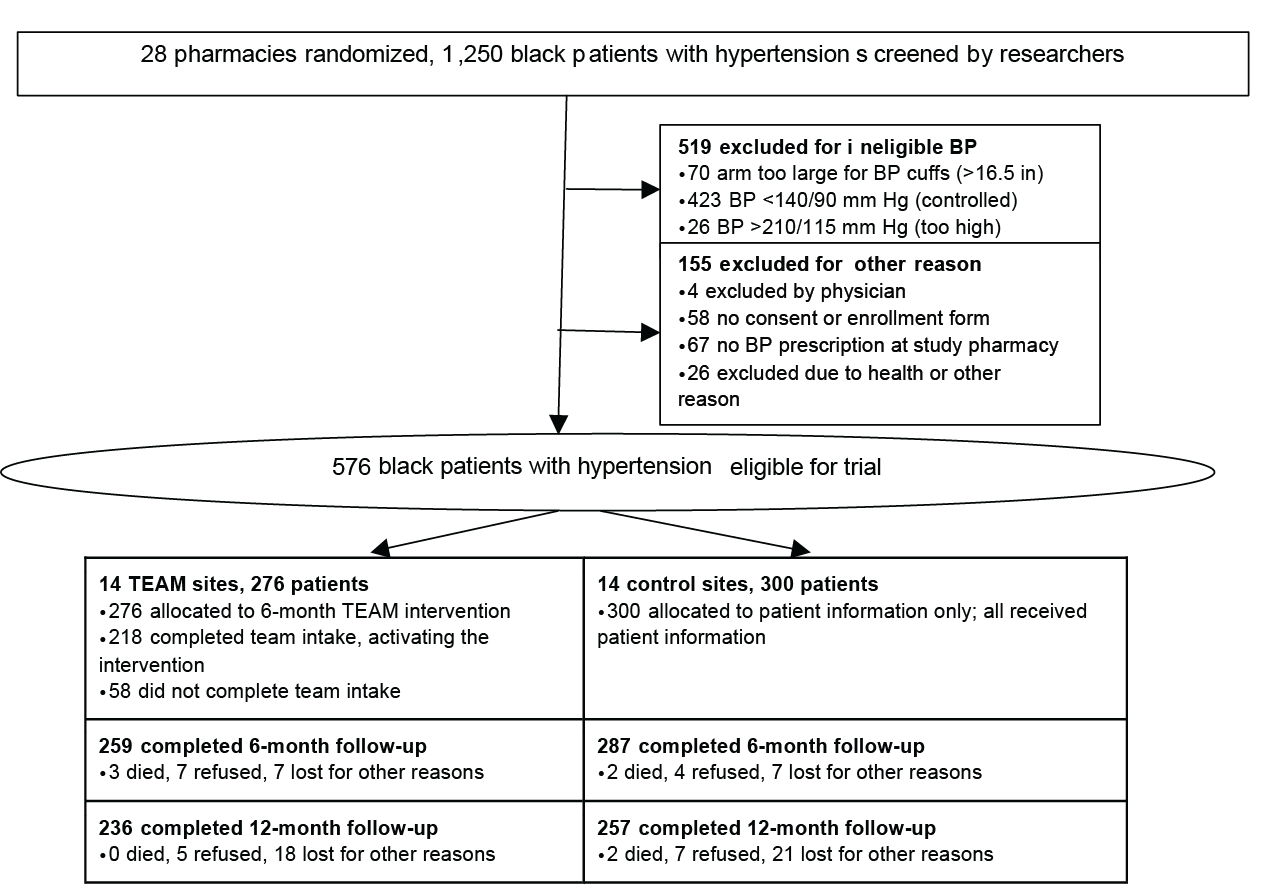
Study Design and Patient flow in TEAM trial*
Outline version of this figure provided below
Source: Svarstad BL, Kotchen JM, Shireman TI et al. Improving refill adherence and hypertension control in black patients: Wisconsin TEAM Trial. JAPhA. 2013;53:524.
*Abbreviations used: BP: blood pressure; TEAM: Team Education and Adherence Monitoring.
This is an accordion element with a series of buttons that open and close related content panels.
Outline Description of Study Design & Patient Flow figure
Study Design and Patient Flor in TEAM Trial
- 28 pharmacies randomized. 1,250 black patients with hypertension screened by researchers.
- 519 patients excluded for ineligible blood pressure
- 70 arm too large for blood pressure cuffs (>16.5 inches)
- 423 blood pressure <140/90 mm HG (controlled)
- 26 blood pressure >210/115 mm Hg (too high)
- 155 excluded for other reason
- 4 excluded by physician
- 58 had no consent or enrollment form
- 67 had no blood pressure prescription at study pharmacy
- 26 excluded due to health or other reason
- 519 patients excluded for ineligible blood pressure
- 567 black patients with hypertension eligible for trial. Patients were split across TEAM sites and control sites.
- TEAM sites:
- 14 TEAM sites with 267 patients
- 276 allocated to 6-month TEAM intervention
- 218 completed team intake, activating the intervention
- 58 did not complete TEAM intake
- 259 patients completed 6-month follow-up
- 3 died, 7 refused, 7 lost for other reasons
- 236 patients completed 12-month follow-up
- 0 died, 5 refused, 18 lost for other reasons
- 14 TEAM sites with 267 patients
- Control sites:
- 14 control sites with 300 patients
- 300 allocated to patient information only; all received patient information
- 287 patients completed 6-month follow-up
- 2 died, 4 refused, 7 lost for other reasons
- 257 patients completed 12-month follow-up
- 2 died, 7 refused, 21 lost for other reasons
- 14 control sites with 300 patients